Report highlights. Animal testing is often called “pre-clinical testing” or “pre-clinical trials.” The National Research Council recommends an end to animal research, and critics cite the inefficacy of animal testing in addition to the pain and suffering research animals undergo.
- 22 million research animals are estimated to populate U.S. laboratories at any given time; fewer than 1 million are protected under animal welfare laws.
- Animal testing costs taxpayers $14 billion annually.
- Over 30% of animal experiments involve moderate-to-severe suffering.
- Over 98% of drugs tested on animals are never sold in stores.
- Worldwide, there are over 192 million research animals.
Jump to a state: AL | AK | AZ | AR | CA | CO | CT | DE | FL | GA | HI | ID | IL | IN | IA | KS | KY | LA | ME | MD | MA | MI | MN | MS | MO | MT | NE | NV | NH | NJ | NM | NY | NC | ND | OH | OK | OR | PA | PR | RI | SC | SD | TN | TX | UT | VT | VA | WA | WV | WI | WY
Animals Testing Statistics
Over a thousand facilities nationwide test on animals from dogs and cats to nonhuman primates. Laboratories are required to report federally-protected animals to the U.S. government; this accounts for a relatively small percentage of the total estimated population.
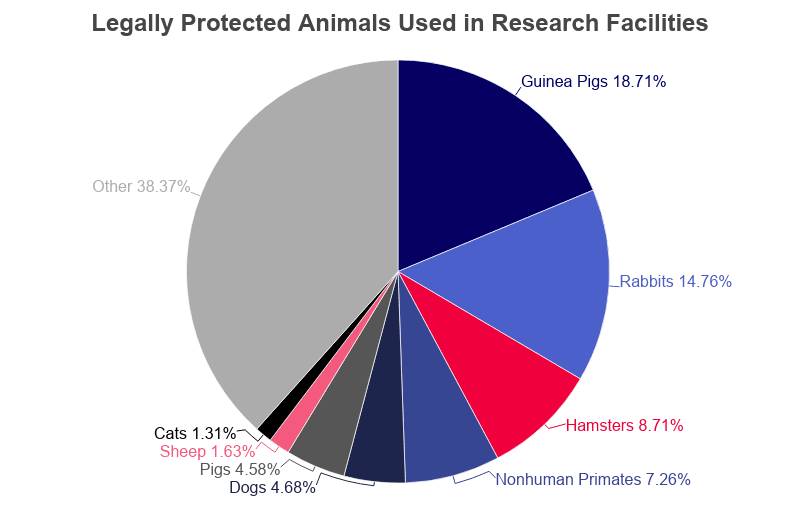
- There are 1,055 active animal testing facilities in the United States.
- These facilities keep a total of 936,675 legally protected animals on site at any given time.
- The average facility houses 888 legally protected animals.
- An average facility may have as many as 20,850 animals on site, including those that are not legally protected (such as certain types of rodents and fish).
- An estimated 7.3% of these animals are housed in federal facilities.
- At least 2.3% are in facilities that perform cosmetic testing.
- 5%-15% of research animals are protected by law.
- As many as 100 million animals may pass through laboratories each year.
| Animal | Nationwide Total | Per Facility |
|---|---|---|
| Cats | 12,266 | 12 |
| Dogs | 43,800 | 42 |
| Guinea Pigs | 175,293 | 166 |
| Hamsters | 81,602 | 77 |
| Nonhuman Primates | 67,972 | 64 |
| Pigs | 42,863 | 41 |
| Rabbits | 138,237 | 131 |
| Sheep | 15,248 | 14 |
| Other Animals | 359,394 | 341 |
| Total | 936,675 | 888 |
Animal Testing in Pharmaceuticals
Pharmaceutical companies invest years of research to put out a viable product. Experimentation reveals how a product may work in multiple scenarios, at multiple concentrations or doses, with a range of test subjects. Toxicity levels differ across species, however, making test results unreliable.
- The pharmaceutical industry is a $1.3 trillion business responsible for 4% of the U.S. Gross Domestic Product.
- 10-15 years is the average time it takes for a drug to complete the development process and be made available to consumers.
- On average, The Food and Drug Administration (FDA) approves 37.75 new drugs per year out of hundreds of applications.
- The number of drugs released to the market increases each year by an average of 14%.
- 92% of drugs tested on animals are deemed ineffective for humans.
- 0.02% of animal-tested drugs are ever made available to the public.
- Animals used in pharmaceutical testing are typically euthanized once the experiment is complete.
Animal Testing in the Federal Government
A number of departments and institutions regulated by the United States Federal Government make use of animal testing in their facilities. U.S. Agencies use animals in testing for many of the same reasons public organizations and private companies do.
- 1.6 million animals, protected and unprotected, are estimated to populate federal laboratories.
- The Department of Agriculture (DOA) researches the health and quality of livestock.
- The Department of Defense (DOD) conducts experiments using technologies and/or chemicals for use in the armed forces.
- The Department of Energy (DOE) tests the effect of new technologies on the health of living creatures.
- The Department of Health and Human Services (DHHS) oversees multiple agencies where testing is done, including the FDA and the National Institute on Drug Abuse (NIDA)
- The Department of the Interior (DOI) uses animal experiments to improve fish and wildlife resource management.
- The Department of Transportation (DOT) conducts research on safe transport of potentially hazardous material and its effect on living creatures.
- The Consumer Product Safety Commission (CPSC) uses animals to test the safety of consumer products.
- The Environmental Protection Agency (EPA) tests the effects of toxic substances on living creatures.
- The National Aeronautics and Space Administration (NASA) experiments on animals to study the effects of flight and space travel on living creatures.
- The Veterans Administration (VA) uses research animals to study treatments for veteran health as well as for education programs.
- Animal testing is a federal requirement for certain research: the FDA, EPA, CPSC, and the Occupational Safety and Health Administration (OSHA) all have processes that explicitly call for animal testing.
Academic Animal Testing
Biomedical research in universities and academic institutions account for a large percentage of experiments using animal test subjects. In academia, ethics of animal research and viability of testing are constantly under question.
- 4% of U.S. medical schools use animal testing.
- Many top universties do not allow animal testing, including Yale, Duke, Harvard, and Tufts.
- Other universities, such as Stanford, have pledged to minimize their use of animal testing.
- Academic animal research may include surgery practices, system examinations, pharmacological testing, psychological tests, and studies of growth.
- 95% of animals used for academic biomedical research are rodents.
Animal Testing in Cosmetics
Experimenting on animals for the purposes of developing cosmetics is the most likely form of animal research to face a ban. Some animal experiments with cosmetics are fatal.
- More than 500,000 animals are used in cosmetic research.
- 250 U.S. companies have petitioned the federal government to put an end to unnecessary cosmetics animal testing.
- 39 countries have banned the use of cosmetics animal testing altogether.
- Animal testing is not a legal requirement for cosmetics approval.
- Common experiments include skin and eye irritation tests, force-feeding, and determination of a product’s “lethal dose.”
Animal Testing Statistics by State
States are required to report the number of federally protected animals they hold in their facilities. Some states may have additional laws regulating animal testing and research, but legislation is minimal.
- Massachusetts reported the highest number of research animals with 91,394, up 1.7% year-over-year (YoY).
- Delaware reported the fewest with 129 federally-protected research animals.
- Delaware also reported the fewest animals per facility, with 65 animals per research center.
- Mississippi has the most crowded laboratories by far, with 10,060 animals per site.
- California has the largest number of research facilities with 121 labs statewide.
| State | Total Animals | Per Facility |
|---|---|---|
| Alabama | 20,939 | 2,327 |
| Alaska | 280 | 70 |
| Arizona | 2,194 | 199 |
| Arkansas | 33,539 | 2,396 |
| California | 54,319 | 449 |
| Colorado | 11,189 | 509 |
| Connecticut | 2,621 | 187 |
| Delaware | 129 | 65 |
| D.C. | 1,966 | 246 |
| Florida | 3,929 | 146 |
| Georgia | 15,084 | 686 |
| Hawaii | 542 | 181 |
| Idaho | 3,675 | 613 |
| Illinois | 7,750 | 235 |
| Indiana | 21,439 | 1,429 |
| Iowa | 18,242 | 829 |
| Kansas | 3,805 | 254 |
| Kentucky | 1,228 | 175 |
| Louisiana | 4,426 | 340 |
| Maine | 61,928 | 6,881 |
| Maryland | 37,224 | 1,201 |
| Massachusetts | 91,394 | 1,428 |
| Michigan | 31,353 | 1,081 |
| Minnesota | 38,013 | 1,462 |
| Mississippi | 60,361 | 10,060 |
| Missouri | 24,395 | 1,061 |
| Montana | 3,412 | 853 |
| Nebraska | 22,807 | 1,901 |
| Nevada | 831 | 166 |
| New Hampshire | 989 | 495 |
| New Jersey | 56,516 | 2,691 |
| New Mexico | 2,026 | 203 |
| New York | 22,867 | 282 |
| North Carolina | 21,136 | 813 |
| North Dakota | 1,074 | 215 |
| Ohio | 61,673 | 1,814 |
| Oklahoma | 8,175 | 584 |
| Oregon | 3,100 | 344 |
| Pennsylvania | 27,620 | 542 |
| Puerto Rico | 4,365 | 1,455 |
| Rhode Island | 827 | 118 |
| South Carolina | 2,416 | 220 |
| South Dakota | 1,640 | 273 |
| Tennessee | 4,340 | 174 |
| Texas | 24,055 | 301 |
| Utah | 3,504 | 350 |
| Vermont | 2,644 | 529 |
| Virginia | 5,457 | 237 |
| Washington | 9,406 | 428 |
| West Virginia | 46,821 | 5,853 |
| Wisconsin | 46,637 | 2,120 |
| Wyoming | 373 | 124 |
Animal Testing in Alabama
There are 9 active animal research labs with an average of 2,327 animals per facility.
- Testing sites hold a total of 20,939 legally protected research animals.
- 124 legally protected research animals are cats.
- 667 protected research animals are dogs.
- 21 research animals are guinea pigs.
- 323 animals are hamsters.
- 259 are nonhuman primates.
- 251 are rabbits.
- 455 are pigs and 29 are sheep.
- 18,810 are other animals.
Animal Testing in Alaska
There are 4 active animal research labs with an average of 70 animals per facility.
- Testing sites hold a total of 280 legally protected research animals.
- 0 legally protected research animals are cats.
- 0 protected research animals are dogs.
- 0 research animals are guinea pigs, hamsters, nonhuman primates, rabbits, pigs or sheep.
- 280 are other animals.
Animal Testing in Arizona
There are 11 active animal research labs with an average of 176 animals per facility.
- Testing sites hold a total of 1,938 legally protected research animals.
- 256 legally protected research animals are cats.
- 432 protected research animals are dogs.
- 9 research animals are guinea pigs.
- 70 animals are hamsters.
- 18 are nonhuman primates.
- 259 are rabbits.
- 330 are pigs and 113 are sheep.
- 707 are other animals.
Animal Testing in Arkansas
There are 14 active animal research labs with an average of 2,394 animals per facility.
- Testing sites hold a total of 33,516 legally protected research animals.
- 23 legally protected research animals are cats.
- 603 protected research animals are dogs.
- 3 research animals are guinea pigs.
- 0 animals are hamsters.
- 78 are nonhuman primates.
- 464 are rabbits.
- 1,192 are pigs and 432 are sheep.
- 30,744 are other animals.
Animal Testing in California
There are 121 active animal research labs with an average of 445 animals per facility.
- Testing sites hold a total of 53,824 legally protected research animals.
- 495 legally protected research animals are cats.
- 1,227 protected research animals are dogs.
- 6,373 research animals are guinea pigs.
- 3,778 animals are hamsters.
- 2,162 are nonhuman primates.
- 17,961 are rabbits.
- 4,393 are pigs and 1,328 are sheep.
- 16,602 are other animals.
Animal Testing in Colorado
There are 22 active animal research labs with an average of 487 animals per facility.
- Testing sites hold a total of 10,721 legally protected research animals.
- 468 legally protected research animals are cats.
- 907 protected research animals are dogs.
- 2,296 research animals are guinea pigs.
- 2,084 animals are hamsters.
- 1 is a nonhuman primate.
- 957 are rabbits.
- 549 are pigs and 561 are sheep.
- 3,366 are other animals.
Animal Testing in Connecticut
There are 14 active animal research labs with an average of 187 animals per facility.
- Testing sites hold a total of 2,612 legally protected research animals.
- 9 legally protected research animals are cats.
- 124 protected research animals are dogs.
- 342 research animals are guinea pigs.
- 251 animals are hamsters.
- 275 are nonhuman primates.
- 299 are rabbits.
- 205 are pigs and 2 are sheep.
- 1,114 are other animals.
Animal Testing in Delaware
There are 2 active animal research labs with an average of 45 animals per facility.
- Testing sites hold a total of 90 legally protected research animals.
- 39 legally protected research animals are cats.
- 60 protected research animals are dogs.
- 0 research animals are guinea pigs, hamsters, or nonhuman primates.
- 18 are rabbits.
- 12 are pigs.
- There are 0 sheep or other animals.
Animal Testing in District of Columbia
There are 8 active animal research labs with an average of 244 animals per facility.
- Testing sites hold a total of 1,952 legally protected research animals.
- 14 legally protected research animals are cats.
- 18 protected research animals are dogs.
- 112 research animals are guinea pigs.
- 94 animals are hamsters.
- 24 are nonhuman primates.
- 58 are rabbits.
- 159 are pigs and 0 are sheep.
- 1,487 are other animals.
Animal Testing in Florida
There are 27 active animal research labs with an average of 135 animals per facility.
- Testing sites hold a total of 3,644 legally protected research animals.
- 285 legally protected research animals are cats.
- 496 protected research animals are dogs.
- 75 research animals are guinea pigs.
- There are no hamsters.
- 1,750 are nonhuman primates.
- 66 are rabbits.
- 316 are pigs and 140 are sheep.
- 801 are other animals.
Animal Testing in Georgia
There are 22 active animal research labs with an average of 665 animals per facility.
- Testing sites hold a total of 14,624 legally protected research animals.
- 460 legally protected research animals are cats.
- 1,131 protected research animals are dogs.
- 820 research animals are guinea pigs.
- 2,141 animals are hamsters.
- 1,908 are nonhuman primates.
- 962 are rabbits.
- 273 are pigs and 69 are sheep.
- 7,320 are other animals.
Animal Testing in Hawaii
There are 3 active animal research labs with an average of 157 animals per facility.
- Testing sites hold a total of 470 legally protected research animals.
- 72 legally protected research animals are cats.
- 189 protected research animals are dogs.
- 0 research animals are guinea pigs, hamsters, or nonhuman primates.
- 11 are rabbits.
- 33 are pigs and 22 are sheep.
- 215 are other animals.
Animal Testing in Idaho
There are 6 active animal research labs with an average of 611 animals per facility.
- Testing sites hold a total of 3,667 legally protected research animals.
- 8 legally protected research animals are cats.
- 7 protected research animals are dogs.
- 0 research animals are guinea pigs, hamsters, nonhuman primates, or pigs.
- 21 are rabbits.
- 901 are sheep.
- 2,736 are other animals.
Animal Testing in Illinois
There are 33 active animal research labs with an average of 217 animals per facility.
- Testing sites hold a total of 7,146 legally protected research animals.
- 604 legally protected research animals are cats.
- 2,459 protected research animals are dogs.
- 184 research animals are guinea pigs.
- 480 animals are hamsters.
- 391 are nonhuman primates.
- 992 are rabbits.
- 1,329 are pigs and 47 are sheep.
- 1,264 are other animals.
Animal Testing in Indiana
There are 15 active animal research labs with an average of 1,417 animals per facility.
- Testing sites hold a total of 21,248 legally protected research animals.
- 191 legally protected research animals are cats.
- 1,622 protected research animals are dogs.
- 502 research animals are guinea pigs.
- 12,735 animals are hamsters.
- 459 are nonhuman primates.
- 1,296 are rabbits.
- 1,163 are pigs and 31 are sheep.
- 3,440 are other animals.
Animal Testing in Iowa
There are 22 active animal research labs with an average of 814 animals per facility.
- Testing sites hold a total of 17,915 legally protected research animals.
- 327 legally protected research animals are cats.
- 304 protected research animals are dogs.
- 650 research animals are guinea pigs.
- 3,243 animals are hamsters.
- 7 are nonhuman primates.
- 471 are rabbits.
- 3,199 are pigs and 711 are sheep.
- 9,330 are other animals.
Animal Testing in Kansas
There are 15 active animal research labs with an average of 217 animals per facility.
- Testing sites hold a total of 3,252 legally protected research animals.
- 553 legally protected research animals are cats.
- 567 protected research animals are dogs.
- 794 research animals are guinea pigs.
- 56 animals are hamsters.
- 10 are nonhuman primates.
- 49 are rabbits.
- 100 are pigs and 12 are sheep.
- 1,664 are other animals.
Animal Testing in Kentucky
There are 7 active animal research labs with an average of 160 animals per facility.
- Testing sites hold a total of 1,121 legally protected research animals.
- 107 legally protected research animals are cats.
- 192 protected research animals are dogs.
- 0 research animals are guinea pigs.
- 40 animals are hamsters.
- 50 are nonhuman primates.
- 20 are rabbits.
- 72 are pigs and 47 are sheep.
- 700 are other animals.
Animal Testing in Louisiana
There are 13 active animal research labs with an average of 330 animals per facility.
- Testing sites hold a total of 4,286 legally protected research animals.
- 140 legally protected research animals are cats.
- 315 protected research animals are dogs.
- 0 research animals are guinea pigs or hamsters.
- 3,341 are nonhuman primates.
- 13 are rabbits.
- 100 are pigs and 0 are sheep.
- 517 are other animals.
Animal Testing in Maine
There are 9 active animal research labs with an average of 6,881 animals per facility.
- Testing sites hold a total of 61,928 legally protected research animals.
- 0 research animals are cats, dogs, guinea pigs, hamsters, or nonhuman primates.
- 6,337 are rabbits.
- 8 are pigs and 27 are sheep.
- 55,556 are other animals.
Animal Testing in Maryland
There are 31 active animal research labs with an average of 1,200 animals per facility.
- Testing sites hold a total of 37,202 legally protected research animals.
- 22 legally protected research animals are cats.
- 502 protected research animals are dogs.
- 4,830 research animals are guinea pigs.
- 5,329 animals are hamsters.
- 8,248 are nonhuman primates.
- 4,475 are rabbits.
- 1,690 are pigs and 44 are sheep.
- 12,084 are other animals.
Animal Testing in Massachusetts
There are 64 active animal research labs with an average of 1,424 animals per facility.
- Testing sites hold a total of 91,123 legally protected research animals.
- 271 legally protected research animals are cats.
- 9,276 protected research animals are dogs.
- 22,629 research animals are guinea pigs.
- 3,276 animals are hamsters.
- 17,282 are nonhuman primates.
- 26,747 are rabbits.
- 5,124 are pigs and 400 are sheep.
- 6,389 are other animals.
Animal Testing in Michigan
There are 29 active animal research labs with an average of 1,076 animals per facility.
- Testing sites hold a total of 31,215 legally protected research animals.
- 138 legally protected research animals are cats.
- 617 protected research animals are dogs.
- 24,107 research animals are guinea pigs.
- 24 animals are hamsters.
- 593 are nonhuman primates.
- 1,351 are rabbits.
- 434 are pigs and 129 are sheep.
- 3,960 are other animals.
Animal Testing in Minnesota
There are 26 active animal research labs with an average of 1,460 animals per facility.
- Testing sites hold a total of 37,966 legally protected research animals.
- 47 legally protected research animals are cats.
- 458 protected research animals are dogs.
- 26,867 research animals are guinea pigs.
- 695 animals are hamsters.
- 173 are nonhuman primates.
- 5,219 are rabbits.
- 3,363 are pigs and 671 are sheep.
- 520 are other animals.
Animal Testing in Mississippi
There are 6 active animal research labs with an average of 10,060 animals per facility.
- Testing sites hold a total of 60,361 legally protected research animals.
- 0 research animals are cats, dogs, guinea pigs, rabbits, or nonhuman primates.
- 29 animals are hamsters.
- 350 are pigs and 24 are sheep.
- 59,958 are other animals.
Animal Testing in Missouri
There are 23 active animal research labs with an average of 1,009 animals per facility.
- Testing sites hold a total of 23,200 legally protected research animals.
- 1,195 legally protected research animals are cats.
- 2,483 protected research animals are dogs.
- 2,997 research animals are guinea pigs.
- 13,191 animals are hamsters.
- 196 are nonhuman primates.
- 1,592 are rabbits.
- 2,072 are pigs and 74 are sheep.
- 595 are other animals.
Animal Testing in Montana
There are 4 active animal research labs with an average of 853 animals per facility.
- Testing sites hold a total of 3,411 legally protected research animals.
- 1 legally protected research animals is a cat.
- 0 research animals are dogs, hamsters, rabbits, or pigs.
- 50 animals are guinea pigs.
- 6 are nonhuman primates.
- 1 is a sheep.
- 3,354 are other animals.
Animal Testing in Nebraska
There are 12 active animal research labs with an average of 1,893 animals per facility.
- Testing sites hold a total of 22,716 legally protected research animals.
- 91 legally protected research animals are cats.
- 208 protected research animals are dogs.
- 5 research animals are guinea pigs.
- 662 animals are hamsters.
- 109 are nonhuman primates.
- 63 are rabbits.
- 2,831 are pigs and 3,024 are sheep.
- 15,813 are other animals.
Animal Testing in Nevada
There are 5 active animal research labs with an average of 153 animals per facility.
- Testing sites hold a total of 764 legally protected research animals.
- 67 legally protected research animals are cats.
- 383 protected research animals are dogs.
- 56 research animals are guinea pigs.
- 0 animals are hamsters, nonhuman primates, or sheep.
- 3 are rabbits.
- 75 are pigs.
- 247 are other animals.
Animal Testing in New Hampshire
There are 2 active animal research labs with an average of 495 animals per facility.
- Testing sites hold a total of 989 legally protected research animals.
- 3 legally protected research animals are hamsters.
- 1 protected research animals is a rabbit.
- 48 research animals are pigs.
- 0 animals are cats, dogs, guinea pigs, nonhuman primates, or sheep.
- 247 are other animals.
Animal Testing in New Jersey
There are 21 active animal research labs with an average of 2,641 animals per facility.
- Testing sites hold a total of 55,459 legally protected research animals.
- 1,057 legally protected research animals are cats.
- 2,583 protected research animals are dogs.
- 18,925 research animals are guinea pigs.
- 22,136 animals are hamsters.
- 1,712 are nonhuman primates.
- 9,143 are rabbits.
- 280 are pigs and 7 are sheep.
- 673 are other animals.
Animal Testing in New Mexico
There are 10 active animal research labs with an average of 200 animals per facility.
- Testing sites hold a total of 1,995 legally protected research animals.
- 31 legally protected research animals are cats.
- 248 protected research animals are dogs.
- 27 research animals are guinea pigs.
- 454 animals are hamsters.
- 685 are nonhuman primates.
- 124 are rabbits.
- 165 are pigs and 0 are sheep.
- 292 are other animals.
Animal Testing in New York
There are 81 active animal research labs with an average of 272 animals per facility.
- Testing sites hold a total of 22,052 legally protected research animals.
- 815 legally protected research animals are cats.
- 1,289 protected research animals are dogs.
- 3,380 research animals are guinea pigs.
- 2,534 animals are hamsters.
- 1,432 are nonhuman primates.
- 2,266 are rabbits.
- 1,018 are pigs and 745 are sheep.
- 9,388 are other animals.
Animal Testing in North Carolina
There are 26 active animal research labs with an average of 798 animals per facility.
- Testing sites hold a total of 20,759 legally protected research animals.
- 377 legally protected research animals are cats.
- 814 protected research animals are dogs.
- 5,425 research animals are guinea pigs.
- 200 animals are hamsters.
- 960 are nonhuman primates.
- 4,925 are rabbits.
- 2,528 are pigs and 177 are sheep.
- 5,730 are other animals.
Animal Testing in North Dakota
There are 5 active animal research labs with an average of 188 animals per facility.
- Testing sites hold a total of 939 legally protected research animals.
- 135 legally protected research animals are cats.
- 149 protected research animals are dogs.
- 0 research animals are guinea pigs.
- 1 animals are hamsters.
- 0 are nonhuman primates.
- 14 are rabbits.
- 11 are pigs and 30 are sheep.
- 734 are other animals.
Animal Testing in Ohio
There are 34 active animal research labs with an average of 1,793 animals per facility.
- Testing sites hold a total of 60,976 legally protected research animals.
- 697 legally protected research animals are cats.
- 1,349 protected research animals are dogs.
- 44,050 research animals are guinea pigs.
- 858 animals are hamsters.
- 538 are nonhuman primates.
- 9,931 are rabbits.
- 1,540 are pigs and 262 are sheep.
- 2,448 are other animals.
Animal Testing in Oklahoma
There are 14 active animal research labs with an average of 569 animals per facility.
- Testing sites hold a total of 7,972 legally protected research animals.
- 203 legally protected research animals are cats.
- 525 protected research animals are dogs.
- 1,646 research animals are guinea pigs.
- 0 animals are hamsters.
- 79 are nonhuman primates.
- 420 are rabbits.
- 62 are pigs and 58 are sheep.
- 5,182 are other animals.
Animal Testing in Oregon
There are 9 active animal research labs with an average of 344 animals per facility.
- Testing sites hold a total of 3,092 legally protected research animals.
- 8 legally protected research animals are cats.
- 28 protected research animals are dogs.
- 127 research animals are guinea pigs.
- 2 animals are hamsters.
- 1,693 are nonhuman primates.
- 29 are rabbits.
- 157 are pigs and 305 are sheep.
- 751 are other animals.
Animal Testing in Pennsylvania
There are 51 active animal research labs with an average of 521 animals per facility.
- Testing sites hold a total of 26,589 legally protected research animals.
- 1,031 legally protected research animals are cats.
- 2,514 protected research animals are dogs.
- 1,223 research animals are guinea pigs.
- 576 animals are hamsters.
- 2,102 are nonhuman primates.
- 13,861 are rabbits.
- 1,232 are pigs and 900 are sheep.
- 4,181 are other animals.
Animal Testing in Puerto Rico
There are 3 active animal research labs with an average of 1,455 animals per facility.
- Testing sites hold a total of 4,364 legally protected research animals.
- 1 legally protected research animals is a cat.
- 18 protected research animals are dogs.
- 0 research animals are guinea pigs or hamsters.
- 4,298 are nonhuman primates.
- 12 are rabbits.
- 9 are pigs and 10 are sheep.
- 17 are other animals.
Animal Testing in Rhode Island
There are 7 active animal research labs with an average of 118 animals per facility.
- Testing sites hold a total of 827 legally protected research animals.
- 0 legally protected research animals are cats.
- 18 protected research animals are dogs.
- 88 research animals are guinea pigs.
- 272 animals are hamsters.
- 32 are nonhuman primates.
- 251 are rabbits.
- 40 are pigs and 4 are sheep.
- 122 are other animals.
Animal Testing in South Carolina
There are 11 active animal research labs with an average of 209 animals per facility.
- Testing sites hold a total of 2,296 legally protected research animals.
- 120 legally protected research animals are cats.
- 176 protected research animals are dogs.
- 119 research animals are guinea pigs.
- 38 animals are hamsters.
- 370 are nonhuman primates.
- 26 are rabbits.
- 95 are pigs and 0 are sheep.
- 1,472 are other animals.
Animal Testing in South Dakota
There are 6 active animal research labs with an average of 273 animals per facility.
- Testing sites hold a total of 1,640 legally protected research animals.
- 0 legally protected research animals are cats, dogs, hamsters, nonhuman primates, or pigs.
- 223 research animals are guinea pigs.
- 6 are rabbits.
- 22 are sheep.
- 1,389 are other animals.
Animal Testing in Tennessee
There are 25 active animal research labs with an average of 164 animals per facility.
- Testing sites hold a total of 4,108 legally protected research animals.
- 232 legally protected research animals are cats.
- 742 protected research animals are dogs.
- 79 research animals are guinea pigs.
- 637 animals are hamsters.
- 74 are nonhuman primates.
- 133 are rabbits.
- 835 are pigs and 17 are sheep.
- 1,591 are other animals.
Animal Testing in Texas
There are 80 active animal research labs with an average of 297 animals per facility.
- Testing sites hold a total of 23,787 legally protected research animals.
- 268 legally protected research animals are cats.
- 1,976 protected research animals are dogs.
- 1,134 research animals are guinea pigs.
- 2,696 animals are hamsters.
- 4,096 are nonhuman primates.
- 2,396 are rabbits.
- 1,896 are pigs and 505 are sheep.
- 9,088 are other animals.
Animal Testing in Utah
There are 10 active animal research labs with an average of 329 animals per facility.
- Testing sites hold a total of 3,292 legally protected research animals.
- 212 legally protected research animals are cats.
- 194 protected research animals are dogs.
- 75 research animals are guinea pigs.
- 1,368 animals are hamsters.
- 14 are nonhuman primates.
- 227 are rabbits.
- 225 are pigs and 174 are sheep.
- 1,015 are other animals.
Animal Testing in Vermont
There are 5 active animal research labs with an average of 517 animals per facility.
- Testing sites hold a total of 2,587 legally protected research animals.
- 57 legally protected research animals are cats.
- 25 protected research animals are dogs.
- 2 research animals are guinea pigs.
- 0 animals are hamsters or nonhuman primates.
- 4 are rabbits.
- 5 are pigs and 2,530 are sheep.
- 21 are other animals.
Animal Testing in Virginia
There are 23 active animal research labs with an average of 237 animals per facility.
- Testing sites hold a total of 5,445 legally protected research animals.
- 12 legally protected research animals are cats.
- 117 protected research animals are dogs.
- 549 research animals are guinea pigs.
- 32 animals are hamsters.
- 43 are nonhuman primates.
- 359 are rabbits.
- 1,360 are pigs and 124 are sheep.
- 2,861 are other animals.
Animal Testing in Washington
There are 22 active animal research labs with an average of 422 animals per facility.
- Testing sites hold a total of 9,293 legally protected research animals.
- 113 legally protected research animals are cats.
- 559 protected research animals are dogs.
- 1,957 research animals are guinea pigs.
- 0 animals are hamsters.
- 3,938 are nonhuman primates.
- 725 are rabbits.
- 275 are pigs and 92 are sheep.
- 1,747 are other animals.
Animal Testing in West Virginia
There are 8 active animal research labs with an average of 5,840 animals per facility.
- Testing sites hold a total of 46,718 legally protected research animals.
- 103 legally protected research animals are cats.
- 93 protected research animals are dogs.
- 0 research animals are guinea pigs, nonhuman primates, or sheep.
- 6 animals are hamsters.
- 44 are rabbits.
- 108 are sheep.
- 46,467 are other animals.
Animal Testing in Wisconsin
There are 22 active animal research labs with an average of 2,086 animals per facility.
- Testing sites hold a total of 45,885 legally protected research animals.
- 752 legally protected research animals are cats.
- 5,096 protected research animals are dogs.
- 2,534 research animals are guinea pigs.
- 1,287 animals are hamsters.
- 8,566 are nonhuman primates.
- 23,380 are rabbits.
- 1,244 are pigs and 226 are sheep.
- 3,552 are other animals.
Animal Testing in Wyoming
There are 3 active animal research labs with an average of 113 animals per facility.
- Testing sites hold a total of 338 legally protected research animals.
- 35 legally protected research animals are cats.
- 38 protected research animals are dogs.
- 8 research animals are guinea pigs.
- 1 animals are hamsters.
- 0 are nonhuman primates.
- 4 are rabbits.
- 11 are pigs and 113 are sheep.
- 163 are other animals.
Use of Different Species
Pre-clinical trials involved animal experimentation require the use of multiple species in order to be considered viable. Research scientists select species based on what they know of the animal’s physiology, how the animal’s physiology may compare to a human’s, what effect they believe the experiment may have on the animal, etc. The animal’s size, metabolism, and medical history are all potential factors when selecting an animal for experimentation.
- For pre-clinical trials to be viable, a minimum of 2 species must be used in experimentation.
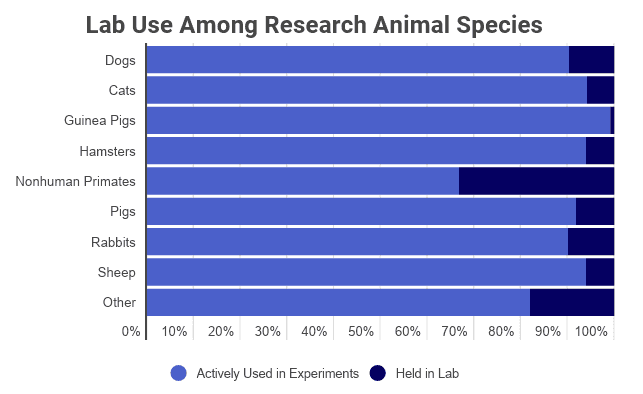
- Among reported animals, guinea pigs are the most widely used, with 171,406 undergoing experimentation every year.
- Rabbits are the second-most used in experiments, followed by Non-human primates.
- The United States uses more primates in experiments than any other country.
- Non-human primates are the most likely species to be held in a research facility without participation in any experiment.
- When used in experiments, non-human primates are most likely to be used to test gastrointestinal disorders.
- Rats are most likely to be used in general and preliminary testing.
Use of Animals in Experiments
The earliest examples of animal experimentation date to the 4th century BC, when animals were dissected to study their anatomy and practice surgeries.
- Test subjects are purposefully exposed to infectious diseases in order to study progression and treatment.
- Other tests include forced ingestion, such as forced feeding or forced inhalation of a toxic substance.
- Animals may be deprived of food and water as part of an experiment.
- Wounds, burns, or other physical damage may be inflicted to study healing.
- Animals are used to study the physiological effects of pain.
- Behavioral studies may induce stress, fear, and panic.
- Genetic manipulation/engineering leaves some animals born with genetic deficiencies, deformities, and potentially fatal disorders.
- Multiple industries use animals in experimentation for varying reasons.
- Animals are used to test cosmetics.
- Biomedical research on animals is common in the pharmaceutical and academic industries.
- Departments and agencies under the federal government use animal testing to predict how new chemicals and technologies might affect humans and/or the environment.
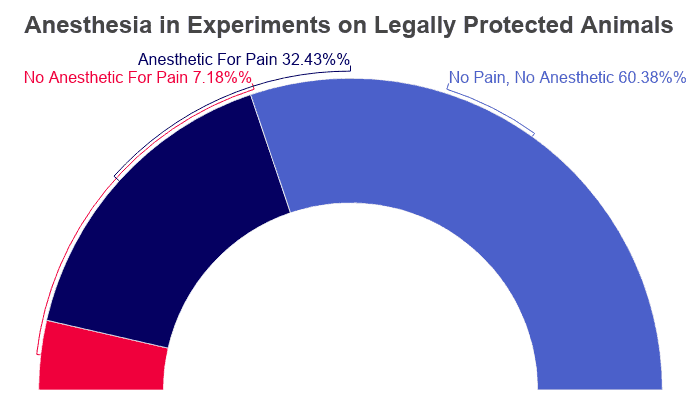
- 471,037 animals reportedly experienced no pain in the course of experimentation.
- 253,002 animals experienced pain and were given pain-killing drugs.
- 56,031 animals experienced pain but were not given any pain-relieving treatment.
- Massachusetts reported the highest rate of pain-free experimentation with 65.3%.
- California reported the highest number of animals given an anesthetic for pain.
- Michigan reported the highest rate of pain and distress among animals during experimentation.
- 21.9% of painful and/or distressing animal experimentation reportedly occurs in Michigan facilities.
- The AWA applies to hospitals, colleges and universities, diagnostic laboratories, pharmaceutical and biotechnology companies, and private research facilities.
- Research facilities are required to form their own Institutional Animal Care and Use Committee to oversee research animal welfare.
- The AWA prohibits researchers from duplicating experiments unnecessarily.
- Critics of the AWA cite its vague language.
- Facilities are required to provide “basic standards of veterinary care and animal husbandry” with little mention of specifics.
- Cages and enclosures must be cleaned every two weeks.
- Potable water must be available twice daily for one hour.
- A caretaker must observe the animals daily.
- Dogs are singled out to be given “the opportunity for exercise.”
- Laboratories are required to “promote the psychological well-being of nonhuman primates.”
- Researchers are obligated to minimize animal suffering “unless withholding such methods is scientifically justified.”
- Still more critics argue the AWA is insufficient for preventing animal suffering.
- APHIS is prohibited from interrupting the conduct of actual research or experimentation.
- Laboratories are not required to report unprotected animals.
- Some animals have no federal protection only if they were bred specifically for research:
- Birds
- Rats of the genus Rattus
- Mice of the genus Mus
- Some animals have no protection under the AWA in any case:
- Coldblooded species
- Fish
- Invertebrates
- 349 state laws regulate use and treatment of animals in research facilities.
- Each state has an average of 7 regulations affecting research animals.
- Michigan offers the most legal protection to research animals with 23 regulations.
- 2 states have zero additional legal protections: Wyoming and Pennsylvania.
- 4 states have banned animal testing for cosmetics: California, New Jersey, New York, and Virginia.
- Scientists already use simulated human organs and tissues as substitutes for human experimentation; these are considered more reliable than testing animals with completely different physiologies.
- Tissue-on-Chips or Organs-on-Chips are human cells that have been manipulated to function as organs and organ systems.
- Computer modeling can simulate chemical effects on a human body based on known parameters, such as toxicity of like-substances.
- Human volunteers are typically given extremely small doses of experimental drugs.
- State-of-the-art scanning and technology can view the inside of the human body with no dissection necessary.
- China is estimated to use the highest number of research animals, with 20.5 million test subjects.
- Japan is the only other country besides China and the United States to test animals in the tens of millions.
- Brussels has banned experiments on cats, dogs, and primates.
- The European Union estimates that only 20% of animal experiments are regulatory requirements.
- 50% of all animal testing in the United Kingdom (UK) is conducted in universities.
- 49% of all animal tests in the UK involved the creation or breeding of genetically altered animals.
- The Chinese government requires animal testing on all cosmetics.
- 52% of Americans oppose the use of animals in research laboratories.
- 21% of Americans oppose genetic engineering of animals due to the potential for suffering.
- Men are 46.8% more likely to support animal research than women.
- People with advanced degrees are 38.4% more accepting of animal research than those with a high school degree as their highest level of educational attainment.
Pain & Treatment in Animal Testing
Animal experimentation is for illnesses or conditions that are painful; these experiments require the animal to experience pain while still others require the animal’s death. If animals are legally protected, research facilities are required to report how many of those protected animals experienced pain, on a scale of mild-to-severe, as part of the experimental process.
Animal Welfare and Research Laws
Federal and state governments specify regulations for use of animals in science and research laboratories. At the federal level, conduct in research and science laboratories is regulated by multiple departments. Animal rights groups, public health officials, universities and private companies also publish reports and guidelines for scientists and researchers in the course of dealing with animals.
Animal Welfare Act
The Animal Welfare Act (AWA) is enforced by the Animal and Plant Health Inspection Service (APHIS) under the United States Department of Agriculture (USDA). Under the AWA, pets have the most rights, whereas some animals have none at all. In research labs, enforcing the AWA is especially difficult.
Health Research Extension Act
The Public Health Service (PHS) Policy on Humane Care and Use of Animals (PHCUA) is a result of the Health Research Extension Act of 1985. This law established guidelines for proper care of animals used in biomedical and behavioral research funded by PHS. The policy itself makes reference to the AWA, noting that PHS regulations are to be considered in conjunction with the rules of the AWA.
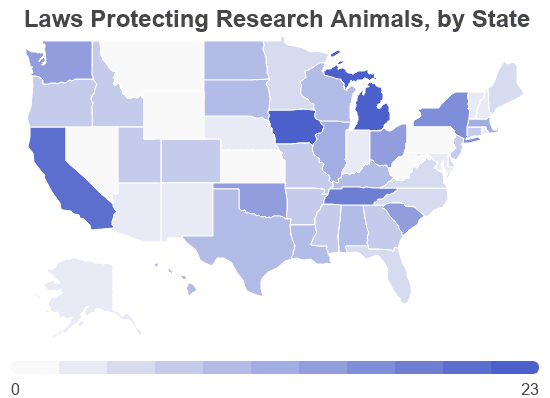
State Legislation
Some states have created laws to fill in gaps left by federal policy. States have recently placed limits on or banned animal testing. Most state anti-cruelty laws exempt research animals, though some supplement these exemptions with additional regulations just for animals involved in laboratory tests.
Alternatives to Animal Research
An increasing number of specialists and institutions call animal testing ineffective and unnecessary. The National Research Council (NRC) of the National Academies of Sciences, Engineers, and Medicine (NASEM) published their official position that animal testing should be discontinued.
International Law and Trends
Worldwide, more countries are placing restrictions on or outright banning animal testing. Industries still make the argument for animal testing in certain cases, however, and some governmental bodies actually require animal testing prior to approval for market release.
Public Opinion
The general public is divided when it comes to animal experimentation and research. Political preferences don’t seem to affect opinion, but the level of education a person received does.
Sources
- USDA Animal Welfare Act
- USDA Animal Welfare Fact Sheet
- Animal Welfare Act Program Information
- ALDF Glossary of Legal Terms
- Animals Used In Research
- Toxicity Testing in the 21st Century: A Vision and a Strategy
- The FDA’s Drug Review Process: Ensuring Drugs are Safe and Effective
- USDA Annual Report Animal Usage in Research 2018
- Preclinical Development
- US Statistics
- Most Americans Accept Genetic Engineering of Animals that Benefits Human Health…
- What is Animal Testing?
- Public Health Service Policy on Humane Care and Use of Laboratory Animals
- Government of Brussels-Capital Bans Animal Tests On Primates, Dogs, And Cats
- Biopharmaceutical Spotlight: The Biopharmaceutical Industry in the United States
- Trends in FDA Drug Approvals Over Last 2 Decades: An Observational Study
- The NIH Microphysiological Systems Program: Tissue-on-Chips for Safety and Efficacy…
- Animal Testing and Medicine
- HSI: About Animal Testing
- Alternatives to Animal Testing
- Use of Laboratory Animals in Biomedical and Behavioral Research
- The Use of Animals in Universities
- Facts and Figures on Animal Testing
- Americans are Divided Over the Use of Animals in Scientific Research
- Cosmetic Testing FAQ
- Ending Cosmetics Animal Testing
